

Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
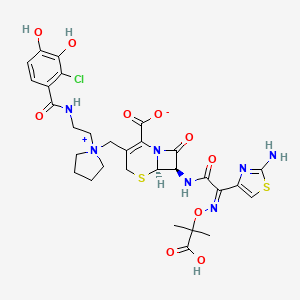

1. S-649266
1. 1225208-94-5
2. Cefiderocol [inn]
3. Gsk2696266
4. Cefiderocol [who-dd]
5. Sz34omg6e8
6. S-649266
7. Cefiderocol (usan)
8. Cefiderocol [usan]
9. (6r,7r)-7-[[(2z)-2-(2-amino-1,3-thiazol-4-yl)-2-(2-carboxypropan-2-yloxyimino)acetyl]amino]-3-[[1-[2-[(2-chloro-3,4-dihydroxybenzoyl)amino]ethyl]pyrrolidin-1-ium-1-yl]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate
10. Pyrrolidinium, 1-(((6r,7r)-7-(((2z)-2-(2-amino-4-thiazolyl)-2-((1-carboxy-1-methylethoxy)imino)acetyl)amino)-2-carboxy-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-en-3-yl)methyl)-1-(2-((2-chloro-3,4-dihydroxybenzoyl)amino)ethyl)-, Inner Salt
11. Rsc 649266
12. Pyrrolidinium, 1-[[(6r,7r)-7-[[(2z)-2-(2-amino-4-thiazolyl)-2-[(1-carboxy-1-methylethoxy)imino]acetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-1-[2-[(2-chloro-3,4-dihydroxybenzoyl)amino]ethyl]-, Inner Salt
13. Cefiderocol [mi]
14. Cefiderocol [usan:inn]
15. Unii-sz34omg6e8
16. Chembl3989974
17. Schembl22508010
18. Dtxsid401098052
19. Akos037648584
20. Db14879
21. Gsk 2696266
22. Bs-14716
23. Hy-17628
24. Cs-0016784
25. D11302
26. S 649266
27. (6r,7r)-7-((2z)-2-(2-amino-1,3-thiazol-4-yl)-2-(((2-carboxypropan-2-yl)oxy)imino)acetamido)-3-((1-(2-(2-chloro-3,4-dihydroxybenzamido)ethyl)pyrrolidin-1-ium-1-yl)methyl)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylate
| Molecular Weight | 752.2 g/mol |
|---|---|
| Molecular Formula | C30H34ClN7O10S2 |
| XLogP3 | 1 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 12 |
| Exact Mass | 751.1497103 g/mol |
| Monoisotopic Mass | 751.1497103 g/mol |
| Topological Polar Surface Area | 310 Ų |
| Heavy Atom Count | 50 |
| Formal Charge | 0 |
| Complexity | 1440 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cefiderocol is indicated for the treatment of complicated urinary tract infections with or without pyelonephritis.
FDA Label
Fetcroja is indicated for the treatment of infections due to aerobic Gram-negative organisms in adults with limited treatment options (see sections 4. 2, 4. 4 and 5. 1).
Consideration should be given to official guidance on the appropriate use of antibacterial agents.
Treatment of infections due to aerobic Gram-negative bacteria
Similarly to other cephalosporins, cefiderocol exerts bactericidal activity against a range of bacterial species. Cefiderocol has primarily shown efficacy against aerobic Gram negative bacteria including *Escherichia coli*, *Klebsiella pneumoniae*, and *Pseudomonas aeruginosa*.
J01D
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01D - Other beta-lactam antibacterials
J01DI - Other cephalosporins and penems
J01DI04 - Cefiderocol
Absorption
A single intravenous dose of 2 g of cefiderocol in healthy patients produces a Cmax of 89.7 mg/L and an AUC of 386 mg\*h/L. In patients with complicated urinary tract infections and a creatinine clearance of at least 60 mL/min, doses of 2 g cefiderocol every 8 hours produced an AUC of 394.7 mg*h/L and a Cmax of 138 mg/L. However the infusion rate for this chronic dosing was 3 times the recommended rate. Cmax and AUC are known to increase proportionally with dosage.
Route of Elimination
98.6% of cefiderocol is eliminated in the urine with 90.6% as the unchanged parent drug. The remaining 8% is eliminated as metabolites. 2.8% is eliminated in the feces. Less than 10% of cefiderocol is metabolized.
Volume of Distribution
Cefiderocol has a mean volume of distribution of 18 L.
Clearance
Cefiderocol has a mean clearance of 5.18 L/h.
Cefiderocol undergoes a small degree of metabolism to a cefiderocol epimer at the 7 position, cefiderocol catechol-3-methoxy and -4-methoxy, and a pyrrolidine chlorobenzamide product (PCBA). PCBA undergoes further metabolism to sulfated, methylated, and glucuronidated metabolites. The enzymes involved in these reactions have yet to be identified and cefiderocol has not been shown to interfere in the metabolism of other agents.
The terminal elimination half-life of cefiderocol is 2-3 h.
Cefiderocol acts by binding to and inhibiting penicillin-binding proteins (PBPs), preventing cell wall synthesis and ultimately causing death of the bacterial cell. Like other -lactam antibiotics cefiderocol is able to enter bacterial cells via passive diffusion through porins. Unlike other -lactams, cefiderocol contains a chlorocatechol group which allows it to chelate iron. Once bound to ferric iron cefiderocol is able to undergo active transport into bacterial cells through iron channels in the outer cell membrane such as those encoded by the *cirA* and *fiu* genes in *E. coli* or the *PiuA* gene in *P. aeruginosa*. Once inside the cell, cefiderocol binds to and inhibits PBP3 with high affinity thereby preventing the linking of peptodoglycan layers via the pentapeptide bridge. PBP1a, 1b, 2,and 4 are also bound and inhibited by cefiderocol but with a lesser potency than PBP3 and are therefore expected to contribute less to its antibacterial effect.
 Jinan Tantu Chemicals offers customized R&D services & production of small molecule APIs & pharmaceutical intermediates.
Jinan Tantu Chemicals offers customized R&D services & production of small molecule APIs & pharmaceutical intermediates.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The collaboration agreement includes provisions to work with ministries of health and other experts to strengthen hospital-based stewardship programmes that ensure appropriate use. These provisions are especially important to avoid fuelling resistance to Fetroja (cefiderocol).
Lead Product(s): Cefiderocol
Therapeutic Area: Infections and Infectious Diseases Brand Name: Fetroja
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Clinton Health Access Initiative
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Collaboration June 15, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Cefiderocol
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Clinton Health Access Initiative
Deal Size : Undisclosed
Deal Type : Collaboration
Details : The collaboration agreement includes provisions to work with ministries of health and other experts to strengthen hospital-based stewardship programmes that ensure appropriate use. These provisions are especial...
Brand Name : Fetroja
Molecule Type : Small molecule
Upfront Cash : Undisclosed
June 15, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
U.S.FDA has approved a supplemental New Drug Application for FETROJA® (cefiderocol) for the treatment of patients 18 years of age or older with hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP).
Lead Product(s): Cefiderocol,Linezolid
Therapeutic Area: Pulmonary/Respiratory Diseases Brand Name: Fetroja
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 28, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Cefiderocol,Linezolid
Therapeutic Area : Pulmonary/Respiratory Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Shionogi Announces FDA Approval of Fetroja® for Hospital-Acquired Bacterial Pneumonia AND Ventila...
Details : U.S.FDA has approved a supplemental New Drug Application for FETROJA® (cefiderocol) for the treatment of patients 18 years of age or older with hospital-acquired bacterial pneumonia and ventilator-associated b...
Brand Name : Fetroja
Molecule Type : Small molecule
Upfront Cash : Not Applicable
September 28, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
FDA has accepted the company’s supplemental New Drug Application (sNDA) for FETROJA® (cefiderocol) and granted Priority Review designation with a Prescription Drug User Fee Act (PDUFA) date of September 27, 2020.
Lead Product(s): Cefiderocol,Linezolid
Therapeutic Area: Pulmonary/Respiratory Diseases Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 01, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Cefiderocol,Linezolid
Therapeutic Area : Pulmonary/Respiratory Diseases
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
FDA Accepts Shionogi’s sNDA with Priority Review for FETROJA® for Hospital-Acquired Pneumonia a...
Details : FDA has accepted the company’s supplemental New Drug Application (sNDA) for FETROJA® (cefiderocol) and granted Priority Review designation with a Prescription Drug User Fee Act (PDUFA) date of September 27, ...
Brand Name : Undisclosed
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 01, 2020

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]2-chloro-3,4-bis((4-methoxybenzyl)oxy)-N-(2-(pyrro...
CAS Number : CAS-1225208-44-5
End Use API : Cefiderocol
About The Company : Jinan Tantu Chemicals Co., Ltd. operates as a Contract Development and Manufacturing Organization (CDMO) that serves pharmaceutical companies worldwide. Our cor...
2-chloro-3,4-bis[(4-methoxyphenyl)methoxy]-Benzald...
CAS Number : CAS-1884263-19-7
End Use API : Cefiderocol
About The Company : Jinan Tantu Chemicals Co., Ltd. operates as a Contract Development and Manufacturing Organization (CDMO) that serves pharmaceutical companies worldwide. Our cor...
7-Amino-3-chloromethyl-3-cephem-4-carboxylic acid ...
CAS Number : CAS-113479-65-5
End Use API : Cefiderocol
About The Company : Jinan Tantu Chemicals Co., Ltd. operates as a Contract Development and Manufacturing Organization (CDMO) that serves pharmaceutical companies worldwide. Our cor...
2-chloro-3,4-bis(4-methoxybenzyloxy)benzoic acid
CAS Number : CAS-137054-46-7
End Use API : Cefiderocol
About The Company : Jinan Tantu Chemicals Co., Ltd. operates as a Contract Development and Manufacturing Organization (CDMO) that serves pharmaceutical companies worldwide. Our cor...
2-(((1-(tert-Butoxy)-2-methyl-1-oxopropan-2-yl)oxy...
CAS Number : 137088-65-4
End Use API : Cefiderocol
About The Company : Jinan Tantu Chemicals Co., Ltd. operates as a Contract Development and Manufacturing Organization (CDMO) that serves pharmaceutical companies worldwide. Our cor...
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
05 Sep 2022
Reply
20 Aug 2022
Reply
12 Dec 2020
Reply
20 Jan 2020
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
