

Synopsis
Synopsis
0
CEP/COS
0
VMF
0
FDF
0
Australia

0
South Africa

0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
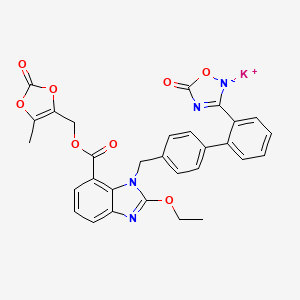

1. 863031-24-7
2. Azilsartan (medoxomil Monopotassium)
3. Azilsartan Kamedoxomil
4. Azilsartan Medoxomil Monopotassium
5. Bcp11249
6. Sb19561
7. Hy-17458
8. Potassium;(5-methyl-2-oxo-1,3-dioxol-4-yl)methyl 2-ethoxy-3-[[4-[2-(5-oxo-1-oxa-4-aza-2-azanidacyclopent-3-en-3-yl)phenyl]phenyl]methyl]benzimidazole-4-carboxylate
9. Potassium 3-(4'-((2-ethoxy-7-(((5-methyl-2-oxo-1,3-dioxol-4-yl)methoxy)carbonyl)-1h-benzo[d]imidazol-1-yl)methyl)-[1,1'-biphenyl]-2-yl)-5-oxo-5h-1,2,4-oxadiazol-2-ide
| Molecular Weight | 606.6 g/mol |
|---|---|
| Molecular Formula | C30H23KN4O8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 10 |
| Exact Mass | 606.11529519 g/mol |
| Monoisotopic Mass | 606.11529519 g/mol |
| Topological Polar Surface Area | 129 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 1110 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
GDUFA
DMF Review : Complete
Rev. Date : 2015-09-10
Pay. Date : 2015-06-26
DMF Number : 29488
Submission : 2015-06-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 29818
Submission : 2015-09-24
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-01-21
Pay. Date : 2020-09-28
DMF Number : 35239
Submission : 2020-09-28
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-10-27
Pay. Date : 2014-08-26
DMF Number : 28452
Submission : 2014-08-14
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2018-01-04
Pay. Date : 2017-08-08
DMF Number : 31895
Submission : 2017-11-28
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 29785
Submission : 2015-09-29
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2019-07-12
Pay. Date : 2019-06-05
DMF Number : 33732
Submission : 2019-04-17
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35932
Submission : 2021-06-30
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-08-06
Pay. Date : 2021-06-23
DMF Number : 28092
Submission : 2014-03-27
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-10-13
Pay. Date : 2021-08-26
DMF Number : 35770
Submission : 2021-03-31
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Registration Number : 305MF10020
Registrant's Address : Ramky Pharma City (SEZ), Plot Nos. 96,97,98,124&126,Parawada-531 019,Visakhapatnam Di...
Initial Date of Registration : 2023-02-08
Latest Date of Registration : 2023-02-08

Registration Number : 305MF10141
Registrant's Address : Jiangxi Fengxin Industrial Park, Fengxin, 330700, Jiangxi Province, PR China
Initial Date of Registration : 2023-12-22
Latest Date of Registration : 2023-12-22

Registration Number : 302MF10016
Registrant's Address : Jiangxi Fengxin Industrial Park, Fengxin, 330700, Jiangxi Province, PR China
Initial Date of Registration : 2020-01-31
Latest Date of Registration : 2021-05-24

Registration Number : 302MF10007
Registrant's Address : 3 Himata, Toyama City, Toyama Prefecture
Initial Date of Registration : 2020-01-10
Latest Date of Registration : 2023-01-11

Registration Number : 301MF10112
Registrant's Address : 174, Sirok-ro, Asan-si, Chungcheongnam-do, KOREA
Initial Date of Registration : 2019-12-25
Latest Date of Registration : 2022-01-20

Azilsartan (for manufacturing only)
Registration Number : 304MF10020
Registrant's Address : 2-3-5 Shimookui, Toyama City, Toyama Prefecture
Initial Date of Registration : 2022-01-26
Latest Date of Registration : 2022-11-16

Registration Number : 302MF10010
Registrant's Address : 1-1 Mikage-cho, Shunan City, Yamaguchi Prefecture
Initial Date of Registration : 2020-01-17
Latest Date of Registration : 2021-05-10

Registration Number : 301MF10062
Registrant's Address : Plot No. 564/A/22, Road No. 92, Jubilee Hills, Hyderabad-500096, Telangana, India
Initial Date of Registration : 2019-09-11
Latest Date of Registration : 2019-09-11

Registration Number : 304MF10129
Registrant's Address : Jiangkou Development Zone, Huangyan, Taizhou City, Zhejiang Province, People's Republ...
Initial Date of Registration : 2022-09-08
Latest Date of Registration : 2022-09-08

Registration Number : 305MF10041
Registrant's Address : Jiangkou Development Zone, Huangyan, Taizhou City, Zhejiang Province, People's Republ...
Initial Date of Registration : 2023-03-29
Latest Date of Registration : 2023-03-29

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Azilsartan Medoxomil Potassium salt IH
Date of Issue : 2022-08-11
Valid Till : 2025-06-26
Written Confirmation Number : WC-0054
Address of the Firm : Block No. 82/B, ECP Road, At & Post. Karakhadi, Tal-Padra, City Karakhadi -39145...
Azilsartan Medoxomil Monopotassium (IH)
Date of Issue : 2022-06-17
Valid Till : 2025-07-02
Written Confirmation Number : WC-0082
Address of the Firm : API Division Kharakhadi, Plot No. 842-843, Kharakhadi, Padra Vadodara-391 450, G...

Azilsartan Medoxomil Potassium IH
Date of Issue : 2022-06-24
Valid Till : 2025-07-02
Written Confirmation Number : WC-0097
Address of the Firm : Block No. 251/P, 252/P, 253 to 255, 256/P,258/P, 276/P, 277/P, 278/P, 279 To 282...

Azilsartan Medoxomil Potassium IH
Date of Issue : 2019-09-27
Valid Till : 2022-07-02
Written Confirmation Number : WC-0097A3
Address of the Firm : Block No. 251/P, 252/P, 253 to 255, 256/P,258/P, 276/P, 277/P, 278/P, 279 To 282...

Date of Issue : 2019-07-26
Valid Till : 2022-05-05
Written Confirmation Number : WC-0362
Address of the Firm : Unit-III Plot No.4 Hetero Infrastructure SEZ N.Narasapuram Village, Nakkapally m...

Date of Issue : 2022-11-18
Valid Till : 2025-09-30
Written Confirmation Number : WC-0455
Address of the Firm : Sy No 14, IDA Gaddapotharm, Jinnaram, Sangareddy (DT), TS-502319

Azilsartan Medoxomil Potassium (IH)
Date of Issue : 2022-04-04
Valid Till : 2025-01-13
Written Confirmation Number : WC-0331
Address of the Firm : Plot No. 38, Rafaleshwar, GIDC, N. H. No 8/A, At & post Jambudia - 363 642 Tal: ...

Date of Issue : 2022-06-17
Valid Till : 2025-07-14
Written Confirmation Number : WC-0021n
Address of the Firm : Sy. Nos: 317, 320, 321, 322, 323, 604 & 605, Rudraram (Village), Patancheru (Man...

Azilsartan Medoxomil Potassium IH
Date of Issue : 2022-06-15
Valid Till : 2025-07-07
Written Confirmation Number : WC-204
Address of the Firm : C-4 Thane, MIDC,belapur road, Vashi, Navi Mumbai,Maharastra, India

Azilsartan Medoxomil Potassium IH
Date of Issue : 2021-06-30
Valid Till : 2022-07-07
Written Confirmation Number : WC-0204A3
Address of the Firm : C-4 Thane, MIDC,belapur road, Vashi, Navi Mumbai, Maharastra

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Azilsartan medoxomil potassium
Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2020-07-09
Registration Number : Number 434-15-ND
Manufacturer Name : Ami Lifesciences Private Lim...
Manufacturer Address : Block No. 82/B, ECP Road At & Post. Karakhadi, Tal-Padra, City: Karakhadi-391 450, Di...
Azilsartan medoxomil potassium
Registrant Name : SG Biochem Co., Ltd.
Registration Date : 2021-07-13
Registration Number : Number 434-15-ND(1)
Manufacturer Name : Ami Lifesciences Private Lim...
Manufacturer Address : Block No. 82/B, ECP Road At & Post. Karakhadi, Tal-Padra, City : Karakhadi-391450, Di...
Azilsartan medoxomil potassium
Registrant Name : Pharmapia Co., Ltd.
Registration Date : 2021-07-14
Registration Number : Wed258-13-ND
Manufacturer Name : CTX Life Sciences Pvt. Ltd.
Manufacturer Address : Block No. 251/P, 252/P, 253 to 255, 256/P, 258/P, 276/P, 277, 278/P, 279 to 282, 283/...

Azilsartan medoxomil potassium
Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2023-10-04
Registration Number : Wed21-18-ND(2)
Manufacturer Name : Honor Lab Limited (Unit-III)
Manufacturer Address : Plot No.4, Hetero Infrastructure SEZ Ltd, N. Narasapuram Village, Nakkapalli Mandal, ...

Azilsartan medoxomil potassium
Registrant Name : Inist ST Co., Ltd.
Registration Date : 2023-07-07
Registration Number : 2220-15-ND
Manufacturer Name : Inist ST Co., Ltd.
Manufacturer Address : 500 Sinnae-ro, Geumwang-eup, Eumseong-gun, Chungcheongbuk-do

Registrant Name : Inist ST Co., Ltd.
Registration Date : 2022-12-16
Registration Number : 2220-14-ND
Manufacturer Name : Inist ST Co., Ltd.
Manufacturer Address : 500 Sinnae-ro, Geumwang-eup, Eumseong-gun, Chungcheongbuk-do

Azilsartan medoxomil potassium
Registrant Name : Korea Biochem Pharmaceutical Co., Ltd.
Registration Date : 2024-01-10
Registration Number : 2152-22-ND
Manufacturer Name : Korea Biochem Pharmaceutical...
Manufacturer Address : 204, Sandan-gil, Jeonui-myeon, Sejong Special Self-Governing City@Plot No.2, APIIC In...

Registrant Name : Kyungbo Pharmaceutical Co., Ltd.
Registration Date : 2021-08-27
Registration Number : 1673-45-ND
Manufacturer Name : Kyungbo Pharmaceutical Co., ...
Manufacturer Address : 174, Silok-ro, Asan-si, Chungcheongnam-do

Registrant Name : Kyungbo Pharmaceutical Co., Ltd.
Registration Date : 2022-04-27
Registration Number : 1673-50-ND
Manufacturer Name : Kyungbo Pharmaceutical Co., ...
Manufacturer Address : 174, Silok-ro, Asan-si, Chungcheongnam-do

Registrant Name : Shinpoong Pharmaceutical Co., Ltd.
Registration Date : 2023-08-09
Registration Number : 1414-9-ND
Manufacturer Name : Shinpoong Pharmaceutical Co....
Manufacturer Address : 7, Wonsi-ro, Danwon-gu, Ansan-si, Gyeonggi-do

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Methyl2-Ethoxy-1-((2'-(N-Hydroxycarbamimidoyl)-[1,...
CAS Number : 147403-65-4
End Use API : Azilsartan
About The Company : Established in May 2012, Shandong Loncom Pharmaceutical operates as a fully owned subsidiary of Shandong Bestcomm Pharmaceutical Co., Ltd. Situated in the Qihe ...
Methyl-2-Ethoxy-1-[[2’- Hydroxycarbamimidoyl) Bi...
CAS Number : CAS-147403-65-4
End Use API : Azilsartan
About The Company : Almelo are the industry leaders in manufacturing advanced intermediates, Active Pharmaceutical Ingredients (APIs) and specialty fine chemicals. With a diverse p...

Methyl 1-[(2'-Cyanobiphenyl-4- yl) Methyl]-2-Ethox...
CAS Number : CAS-139481-44-0
End Use API : Azilsartan
About The Company : Almelo are the industry leaders in manufacturing advanced intermediates, Active Pharmaceutical Ingredients (APIs) and specialty fine chemicals. With a diverse p...

2-Ethoxy-1-[(2-5-Oxo-4,5- Dihydro-1,2,4-Oxadiazol-...
CAS Number : CAS-147403-03-0
End Use API : Azilsartan
About The Company : Almelo are the industry leaders in manufacturing advanced intermediates, Active Pharmaceutical Ingredients (APIs) and specialty fine chemicals. With a diverse p...

1H-Benzimidazole-7-Carboxylic Acid-1-[[2'-(2, 5-Di...
CAS Number : CAS-147403-52-9
End Use API : Azilsartan
About The Company : Almelo are the industry leaders in manufacturing advanced intermediates, Active Pharmaceutical Ingredients (APIs) and specialty fine chemicals. With a diverse p...

1H-Benzimidazole-7-carboxylic acid, 2-ethoxy-1-[[2...
CAS Number : 147403-65-4
End Use API : Azilsartan
About The Company : CTX is one of the youngest and fastest-growing Indian API and Intermediate manufacturing companies doing business in over 70 countries. In these five years, all...

1H-Benzimidazole-7-carboxylic acid, 1-[[2'-(2,5-di...
CAS Number : 147403-52-9
End Use API : Azilsartan
About The Company : CTX is one of the youngest and fastest-growing Indian API and Intermediate manufacturing companies doing business in over 70 countries. In these five years, all...

1H-Benzimidazole-7-carboxylic acid, 1-[[2'-(2,5-d...
CAS Number : 147403-03-0
End Use API : Azilsartan
About The Company : CTX is one of the youngest and fastest-growing Indian API and Intermediate manufacturing companies doing business in over 70 countries. In these five years, all...

1-(2-cyanobiphenyl-4-yl-methyl)- 2-ethoxybenzimida...
CAS Number : 139481-44-0
End Use API : Azilsartan
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

1 -[[2-(2,5-dihydro-5 -oxo-1,2,4-oxadiazol-3 -yl)[...
CAS Number : 147403-52-9
End Use API : Azilsartan
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 40mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
Brand Name : EDARBI
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 40MG MEDOXOMIL
Packaging :
Approval Date : 2011-02-25
Application Number : 200796
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : RX
Registration Country : USA
AZILSARTAN KAMEDOXOMIL; CHLORTHALIDONE
Brand Name : EDARBYCLOR
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 40MG MEDOXOMIL;25MG
Packaging :
Approval Date : 2011-12-20
Application Number : 202331
Regulatory Info : RX
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Canada
AZILSARTAN MEDOXOMIL (AZILSARTAN MEDOXOMIL POTASSIUM)
Brand Name : EDARBYCLOR
Dosage Form : TABLET
Dosage Strength : 40MG
Packaging :
Approval Date :
Application Number : 2397749
Regulatory Info : Prescription
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : DISCN
Registration Country : USA
Brand Name : AZILSARTAN MEDOXOMIL
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 80MG MEDOXOMIL
Packaging :
Approval Date : 2022-07-20
Application Number : 214489
Regulatory Info : DISCN
Registration Country : USA

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Edarbi
Dosage Form : TABLET
Dosage Strength : 20 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Ipreziv
Dosage Form : TABLET
Dosage Strength : 80 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 40mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 80mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Azilsartanum medoxomilum; Chlortalidonum
Brand Name : Edarbyclor
Dosage Form : Filmtabl
Dosage Strength : 40/12.5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Edarbi
Dosage Form : TABLET
Dosage Strength : 80 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Edarbi
Dosage Form : TABLET
Dosage Strength : 40 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Edarbi
Dosage Form : TABLET
Dosage Strength : 20 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Ipreziv
Dosage Form : TABLET
Dosage Strength : 80 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Ipreziv
Dosage Form : TABLET
Dosage Strength : 40 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 20mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 20mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 80mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Edarbi
Dosage Form : Tablet
Dosage Strength : 80mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Azilsartanum medoxomilum; Chlortalidonum
Brand Name : Edarbyclor
Dosage Form : Filmtabl
Dosage Strength : 40/12.5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
