

Synopsis
Synopsis
0
VMF
DRUG PRODUCT COMPOSITIONS
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
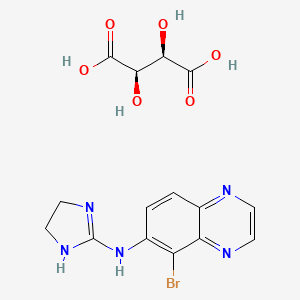

1. 5-bromo-6-(2-imidazolin-2-ylamino)quinoxaline D-tartrate
2. 5-bromo-6-(imidazolidinylideneamino)quinoxaline
3. 5-bromo-6-(imidazolin-2-ylamino)quinoxaline
4. Agn 190342
5. Agn-190342
6. Agn190342
7. Alphagan
8. Alphagan P
9. Brimonidine
10. Brimonidine Purite
11. Brimonidine Tartrate (1:1)
12. Brimonidine Tartrate (1:1), (s-(r*,r*))-isomer
13. Brimonidine Tartrate, (r-(r*,r*))-isomer
14. Bromoxidine
15. Mirvaso
16. Ratio Brimonidine
17. Ratio-brimonidine
18. Sanrosa
19. Uk 14,304
20. Uk 14,304 18
21. Uk 14,304-18
22. Uk 14,30418
23. Uk 14,308
24. Uk 14304
25. Uk 14308
26. Uk-14,304-18
27. Uk-14,308
28. Uk-14304
29. Uk14,30418
30. Uk14,308
31. Uk14304
1. 70359-46-5
2. Alphagan
3. Brimonidine Tartarate
4. Brimonidine L-tartrate
5. Lumify
6. Brimonidinne Tartrate
7. 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)quinoxalin-6-amine (2r,3r)-2,3-dihydroxysuccinate
8. Mirvaso
9. 4s9cl2dy2h
10. Agn 190342-lf
11. Brimonidine D-tartarate
12. 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)quinoxalin-6-amine;(2r,3r)-2,3-dihydroxybutanedioic Acid
13. Uk-14304-18
14. Qoliana
15. Agn-190342-lf
16. Bromoxidine Tartrate
17. (5-bromo-quinoxalin-6-yl)-(4,5-dihydro-1h-imidazol-2-yl)-amine L-tartrate
18. Brimonidine Purite
19. Brimonidine Tartrate [usan]
20. (2r,3r)-2,3-dihydroxybutanedioic Acid; 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)quinoxalin-6-amine
21. 6-quinoxalinamine, 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)-, (2r,3r)-2,3-dihydroxybutanedioate (1:1)
22. 6-quinoxalinamine, 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)-, (r-(r*,r*))-2,3-dihydroxybutanedioate (1:1)
23. 59803-99-5
24. Ocu300
25. Unii-4s9cl2dy2h
26. Ocu-300
27. Agn-190342lf
28. Alphagan-p
29. Cd-07805
30. Agn 190342lf
31. Brimonidine Tartrate [usan:jan]
32. N-(5-bromoquinoxalin-6-yl)imidazolidin-2-imine;(2r,3r)-2,3-dihydroxybutanedioic Acid
33. Alphagan (tn)
34. 304 Tartrate
35. Uk-1430418
36. 5-bromo-n-
37. Uk14304 Tartrate
38. Agn190342 Tartrate
39. Uk 14304 (tartrate)
40. Agn190342 (tartrate)
41. Uk 14,304 (tartrate)
42. Schembl265607
43. Chembl1200389
44. Ex-a5415a
45. Hy-b0659a
46. Dtxsid70911371
47. Brimonidine Tartrate (jan/usan)
48. Brimonidine Tartrate [jan]
49. Hms3715p18
50. Act08633
51. Brimonidine D-tartrate [mi]
52. Brimonidine Tartrate [vandf]
53. Brimonidine Tartrate [mart.]
54. Mfcd07773072
55. Brimonidine Tartrate [usp-rs]
56. Brimonidine Tartrate [who-dd]
57. Akos016845265
58. Ccg-221278
59. Cs-4496
60. 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl)quinoxalin-6-amine(2r,3r)-2,3-dihydroxysuccinate
61. As-18083
62. Brimonidine Tartrate [orange Book]
63. Brimonidine Tartrate [ep Monograph]
64. Brimonidine Tartrate [usp Monograph]
65. Cd07805/47
66. Cd-07805/47
67. Combigan Component Brimonidine Tartrate
68. A19740
69. D02076
70. Simbrinza Component Brimonidine Tartrate
71. Brimonidine Tartrate Component Of Combigan
72. 359b465
73. Brimonidine Tartrate Component Of Simbrinza
74. (4,5-bihydro-1h-imidazol-2-yl)quinoxalin-6-amine L-tartrate
75. 5-bromo-n-(4,5-dihydro-1h-imidazol-2-yl) Quinoxalin-6-amine Tartrate
76. 109826-56-4
| Molecular Weight | 442.22 g/mol |
|---|---|
| Molecular Formula | C15H16BrN5O6 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 5 |
| Exact Mass | 441.02840 g/mol |
| Monoisotopic Mass | 441.02840 g/mol |
| Topological Polar Surface Area | 177 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 442 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Brimonidine tartrate |
| Drug Label | Brimonidine Tartrate Ophthalmic Solution, 0.2% is a relatively selective alpha-2 adrenergic agonist for ophthalmic use. The chemical name of brimonidine tartrate is 5-bromo-6 (2-imidazolidinylideneamino) quinoxaline L-tartrate. It is a white to sligh... |
| Active Ingredient | Brimonidine tartrate |
| Dosage Form | Solution/drops; Solution |
| Route | ophthalmic; Ophthalmic |
| Strength | 0.2%; 0.1% |
| Market Status | Tentative Approval; Prescription |
| Company | Apotex; Bausch And Lomb; Alcon Pharms; Sandoz; Akorn |
| 2 of 2 | |
|---|---|
| Drug Name | Brimonidine tartrate |
| Drug Label | Brimonidine Tartrate Ophthalmic Solution, 0.2% is a relatively selective alpha-2 adrenergic agonist for ophthalmic use. The chemical name of brimonidine tartrate is 5-bromo-6 (2-imidazolidinylideneamino) quinoxaline L-tartrate. It is a white to sligh... |
| Active Ingredient | Brimonidine tartrate |
| Dosage Form | Solution/drops; Solution |
| Route | ophthalmic; Ophthalmic |
| Strength | 0.2%; 0.1% |
| Market Status | Tentative Approval; Prescription |
| Company | Apotex; Bausch And Lomb; Alcon Pharms; Sandoz; Akorn |
Mirvaso is indicated for the symptomatic treatment of facial erythema of rosacea in adult patients.
Adrenergic alpha-2 Receptor Agonists
Compounds that bind to and activate ADRENERGIC ALPHA-2 RECEPTORS. (See all compounds classified as Adrenergic alpha-2 Receptor Agonists.)
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
D11AX21
 Farmak works in the development, production and marketing of APIs, Intermediates & Specialties// FDA inspected.
Farmak works in the development, production and marketing of APIs, Intermediates & Specialties// FDA inspected.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2012-10-26
Pay. Date : 2012-12-19
DMF Number : 15585
Submission : 2001-08-15
Status : Active
Type : II

Certificate Number : CEP 2014-331 - Rev 03
Issue Date : 2024-03-06
Type : Chemical
Substance Number : 2760
Status : Valid

Registration Number : 302MF10029
Registrant's Address : Na vlcinci 16/3 Klasterni Hradisko 779 00 Olomouc Czech Republic
Initial Date of Registration : 2020-02-13
Latest Date of Registration : --

NDC Package Code : 63278-0488
Start Marketing Date : 2002-01-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Hiple Co., Ltd.
Registration Date : 2013-12-20
Registration Number : 20131220-200-I-380-02
Manufacturer Name : Farmak as
Manufacturer Address : Na Vlcinci 16/3, Klasterni Hradisko, 779 00 Olomouc, Czech Republic
 ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.

GDUFA
DMF Review : Reviewed
Rev. Date : 2012-12-26
Pay. Date : 2012-11-23
DMF Number : 15601
Submission : 2001-08-27
Status : Active
Type : II

Certificate Number : R1-CEP 2013-353 - Rev 00
Issue Date : 2019-04-24
Type : Chemical
Substance Number : 2760
Status : Valid

NDC Package Code : 53296-0050
Start Marketing Date : 2014-09-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (75kg/75kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Hansol Pharma Co., Ltd.
Registration Date : 2024-05-01
Registration Number : 20141201-200-I-451-04(1)
Manufacturer Name : MEDICHEM S.A.
Manufacturer Address : Pirineus street, 127, Poligon Industrial de Celra, 17460 Celra (Girona), Spain
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.

GDUFA
DMF Review : Reviewed
Rev. Date : 2012-11-23
Pay. Date : 2012-12-03
DMF Number : 22510
Submission : 2009-01-28
Status : Active
Type : II

Certificate Number : CEP 2022-161 - Rev 00
Issue Date : 2024-02-02
Type : Chemical
Substance Number : 2760
Status : Valid

NDC Package Code : 82133-104
Start Marketing Date : 2023-12-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25051
Submission : 2011-07-05
Status : Active
Type : II
Date of Issue : 2019-07-29
Valid Till : 2022-07-28
Written Confirmation Number : WC-0063
Address of the Firm : Plot No. 99, MIDC Area, Dhatav, Roha, Dist Raigad

NDC Package Code : 53747-036
Start Marketing Date : 2011-07-05
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22230
Submission : 2008-12-01
Status : Active
Type : II

Certificate Number : R0-CEP 2018-124 - Rev 00
Issue Date : 2020-05-14
Type : Chemical
Substance Number : 2760
Status : Valid
Date of Issue : 2019-10-07
Valid Till : 2022-09-08
Written Confirmation Number : WC-0072
Address of the Firm : Plot No. 25/B, Phase-III, I.D.A., Jeedimetla, Hyderabad-500 055. AP.

NDC Package Code : 66022-0110
Start Marketing Date : 2013-08-23
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 29 companies offering QOLIANA
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
