

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF
0
FDA Orange Book

0
Canada

0
Australia

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
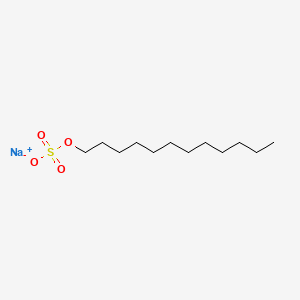

1. Dodecyl Sulfate, Sodium
2. Irium
3. Lauryl Sulfate, Sodium
4. Sodium Lauryl Sulfate
5. Sulfate, Sodium Dodecyl
6. Sulfate, Sodium Lauryl
1. 151-21-3
2. Sodium Lauryl Sulfate
3. Sodium Dodecylsulfate
4. Sodium Lauryl Sulphate
5. Sodium Dodecyl Sulphate
6. Dodecyl Sodium Sulfate
7. Neutrazyme
8. Sodium N-dodecyl Sulfate
9. Irium
10. Sulfuric Acid Monododecyl Ester Sodium Salt
11. Dodecyl Sulfate Sodium Salt
12. Dodecyl Sulfate, Sodium Salt
13. Anticerumen
14. Duponal
15. Duponol
16. Gardinol
17. Sodium Monododecyl Sulfate
18. Dreft
19. Aquarex Methyl
20. Duponol Methyl
21. Solsol Needles
22. Stepanol Methyl
23. Duponol Waqa
24. Stepanol Wac
25. Stepanol Waq
26. Duponol Qx
27. Richonol Af
28. Perlandrol L
29. Perlankrol L
30. Sipex Sb
31. Sipex Sd
32. Standapol Wa-ac
33. Stepanol Me Dry
34. Duponol Me
35. Richonol A
36. Richonol C
37. Sintapon L
38. Duponol C
39. Maprofix Lk
40. Standapol Waq
41. Stepanol Me
42. Stepanol Wa
43. Akyposal Sds
44. Carsonol Sls
45. Maprobix Neu
46. Maprofix Neu
47. Maprofix Wac
48. Aquarex Me
49. Dupanol Waq
50. Duponol Qc
51. Duponol Wa
52. Duponol Wa Dry
53. Duponol Waq
54. Empicol Lpz
55. Hexamol Sls
56. Melanol Cl
57. Duponal Waqe
58. Duponol Waqe
59. Duponol Waqm
60. Lanette Wax-s
61. Sterling Wa Paste
62. Conco Sulfate Wa
63. Conco Sulfate Wn
64. Nikkol Sls
65. Orvus Wa Paste
66. Sipex Op
67. Sipex Sp
68. Sipex Ub
69. Sipon Ls
70. Sipon Pd
71. Sipon Wd
72. Detergent 66
73. Montopol La Paste
74. Sipon Lsb
75. Maprofix Wac-la
76. Sterling Waq-ch
77. Cycloryl 21
78. Cycloryl 31
79. Stepanol Wa Paste
80. Conco Sulfate Wag
81. Conco Sulfate Wan
82. Conco Sulfate Was
83. Quolac Ex-ub
84. Odoripon Al 95
85. Sodiumdodecylsulfate
86. Avirol 118 Conc
87. Cycloryl 580
88. Cycloryl 585n
89. Lauyl Sodium Sulfate
90. Maprofix 563
91. Sinnopon Ls 95
92. Stepanol T 28
93. Sodium Laurilsulfate
94. Steinapol Nls 90
95. Empicol Ls 30
96. Empicol Lx 28
97. Lauryl Sodium Sulfate
98. Melanol Cl 30
99. Nals
100. Rewopol Nls 30
101. Standapol Waq Special
102. Standapol Was 100
103. Sinnopon Ls 100
104. Stepanol Wa-100
105. Carsonol Sls Special
106. Standapol 112 Conc
107. Stepanol Me Dry Aw
108. Avirol 101
109. Emersal 6400
110. Monogen Y 100
111. Carsonol Sls Paste B
112. Sodium;dodecyl Sulfate
113. Stepanol Methyl Dry Aw
114. Berol 452
115. Emal 10
116. Emal O
117. Sipon Ls 100
118. N-dodecyl Sulfate Sodium
119. Sodium Monolauryl Sulfate
120. Monododecyl Sodium Sulfate
121. Sodiumlauryl Ether Sulfate
122. Lauryl Sulfate Sodium Salt
123. Conco Sulfate Wa-1200
124. Conco Sulfate Wa-1245
125. Dehydag Sulfate Gl Emulsion
126. Product No. 75
127. Product No. 161
128. Mfcd00036175
129. Emulsifier No. 104
130. Texapon K 12 P
131. Chebi:8984
132. P And G Emulsifier 104
133. Sodium Lauryl Sulfate Ether
134. Sls
135. Sodium Laurylsulfate
136. Nsc-402488
137. Texapon K 1296
138. Nci-c50191
139. Laurylsulfuric Acid Sodium Salt
140. Natriumalkyl(c8-c20)-sulfate
141. Dodecyl Alcohol, Hydrogen Sulfate, Sodium Salt
142. Dodecylsulfuric Acid Sodium Salt
143. Sodium Lauryl Sulfate, Synthetic
144. Finasol Osr2
145. Incronol Sls
146. Natriumlaurylsulfat
147. 368gb5141j
148. Ncgc00091020-03
149. E487
150. Jordanol Sl-300
151. Finasol Osr(sub 2)
152. Dodecyl Sulfate Sodium
153. Monagen Y 100
154. Perklankrol Esd 60
155. Caswell No. 779
156. Natrium Laurylsulfuricum
157. Dsstox_cid_6031
158. Dsstox_rid_77989
159. Dsstox_gsid_26031
160. 12738-53-3
161. 12765-21-8
162. 1334-67-4
163. Laurylsiran Sodny [czech]
164. Lauryl Sulfate, Sodium Salt
165. Dehydrag Sulfate Gl Emulsion
166. Dehydag Sulphate Gl Emulsion
167. Laurylsiran Sodny
168. Rhodapon Ub
169. Sodium Lauryl Sulfate 30%
170. Cas-151-21-3
171. Ccris 6272
172. Lauryl Sulfate Sodium
173. Hsdb 1315
174. Sodium Lauryl Sulfate, Dental Grade
175. Einecs 205-788-1
176. Epa Pesticide Chemical Code 079011
177. Nsc 402488
178. Cp 75424
179. Empicol
180. Ai3-00356
181. Unii-368gb5141j
182. Sodium Lauryl Sulfate [jan:nf]
183. Sodiumlauryl Sulfate
184. Sulfuric Acid Monododecyl Ester Sodium Salt (1:1)
185. Sodium Dodecylsulphate
186. Sodium Dedecyl Sulfate
187. Sodium-dodecyl-s-sds
188. Ipc-sds
189. Sodium N-dodecyl Sulphate
190. Sodium Lauryl Sulfate Nf
191. Sds (20% Solution)
192. Sodium Monododecyl Sulphate
193. Lauryl Sulphate Sodium Salt
194. Ec 205-788-1
195. Dodecyl Sulphate Sodium Salt
196. Schembl1102
197. Sodium Lauryl Sulfate, Sds
198. Sodium Dodecyl Sulfate (sds)
199. Chembl23393
200. Sodium Dodecyl Sulphate (sds)
201. Sodium Dodecyl Sulphate Solution
202. Dtxsid1026031
203. Dodecyl Sulfuric Acid Sodium Salt
204. Dodecyl Sulphuric Acid Sodium Salt
205. Sodium Lauryl Sulfate (jp17/nf)
206. Sodium Lauryl Sulfate [ii]
207. Sodium Lauryl Sulfate [mi]
208. Bcp30594
209. Cs-b1770
210. Sodium Lauryl Sulfate [fcc]
211. Sodium Lauryl Sulfate [jan]
212. Tox21_111059
213. Tox21_201614
214. Tox21_300149
215. Bdbm50530482
216. Sodium Laurilsulfate [mart.]
217. Sodium Lauryl Sulfate [hsdb]
218. Sodium Lauryl Sulfate [inci]
219. Sodium Lauryl Sulfate [vandf]
220. Akos015897278
221. Akos025147308
222. Tox21_111059_1
223. Db00815
224. Dodecyl Sulfuric Acid Ester Sodium Salt
225. Sodium Lauryl Sulfate [usp-rs]
226. Sodium Lauryl Sulfate [who-dd]
227. Ncgc00091020-01
228. Ncgc00091020-02
229. Ncgc00254225-01
230. Ncgc00259163-01
231. Ncgc00274082-01
232. As-14730
233. Sodium Laurilsulfate [ep Impurity]
234. Lauryl Sulfate, Sodium Salt (25% Aq.)
235. Sodium Laurilsulfate [ep Monograph]
236. D1403
237. Ft-0603358
238. Ft-0700721
239. I0352
240. S0588
241. D01045
242. F16341
243. S-4600
244. S-4601
245. Sodium Dodecyl Sulfate Biotech Grd 100g
246. Q422241
247. Sodium N-dodecyl Sulfate, 98%, For Electrophoresis
248. Sodium N-dodecyl Sulfate (sds), 20% Aqueous Solution
249. F0001-0539
250. Z1695728983
251. Sodium Dodecylsulfate;sodium Lauryl Sulphate;dodecyl Sodium Sulfate
252. Dodecyl Sodium Sulfate, Dodecyl Sulfate Sodium Salt, Lauryl Sulfate Sodium Salt, Sds, Sodium Lauryl Sulfate
| Molecular Weight | 288.38 g/mol |
|---|---|
| Molecular Formula | C12H25NaO4S |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 12 |
| Exact Mass | 288.13712473 g/mol |
| Monoisotopic Mass | 288.13712473 g/mol |
| Topological Polar Surface Area | 74.8 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 249 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
/VET/ foot & mouth disease virus is highly resistant to.../sodium lauryl sulfate/, yet TGE virus is sensitive... fungistatic (incl Candida & Trichophyton spp) & concn of 2% & over eliminated drug resistance & sex transfer factors in E coli. Inhibits growth of many G-pos bacteria...ineffective against G-neg types.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 542
/VET/ Sodium lauryl sulfate is used as a flea and tick repellant in one registered pesticide product--a flea and tick shampoo for cats and dogs. Sodium lauryl sulfate also is a widely used component of many nonpesticidal consumer products currently marketed in the United States, including shampoos and fruit juices.
USEPA/Office of Prevention, Pesticides and Toxic Substances; Registration Eligibility Decision Document - Lauryl Sulfate Salts p. 3 EPA-73B-F-93-Q09 (September 1993) Available from, as of June 16, 2014: https://www.epa.gov/pesticides/reregistration/status.htm
/VET/ as wetting agent for some antibiotics & antimicrobials (tylosin, sulfaquinoxaline, tyrothricin, etc) for oral & topical use. Widely used in ointment bases & as wetting agent for some insecticides & anthelmintics. Also useful in producing clear gel shampoos.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 542
/EXPL THER/ About 1/3 of HIV positive mothers transmit the virus to their newborns, and 1/2 of these infections occur during breastfeeding. Sodium dodecyl sulfate (SDS), an anionic surfactant, is a common ingredient of cosmetic and personal care products. SDS is "readily biodegradable" with low toxicity and "is of no concern with respect to human health". Up to 1 g of SDS/kg is the maximum safe dose for children. Alkyl sulfates, including SDS, are microbicidal against HIV types 1 and 2, herpes simplex virus type 2 (HSV-2), human papillomaviruses and chlamydia. /The study/ hypothesizes that SDS treatment of milk will inactivate HIV-1 without significant harm to its nutritional value and protective functions and may define a treatment of choice for breastwas at 37 degrees C for 10 min. SDS-PAGE and Lowry were used to analyze protein content. Antibody content and function was studied by rocket immunoelectrophoresis (RIE), immunoturbodimentric (ITM) quantitation and ELISA. The creamatocrit was also analyzed. HIV-1 infectivity was measured by MAGI assay. SDS removal was by Detergent-OutN (Geno Technology, Inc.). SDS quantitation is by methylene blue-chloroform method. Inactivation of HIV-1 with SDS occurs at or above 0.025%. In milk samples, 1% and 0.1% SDS reduced HSV-2 infectivity. At least 90% of SDS can be efficiently removed with Detergent-OutN, with protein recovery of 80%-100%. Gross protein species are conserved as indicated by PAGE analyses. Fat and energy content of SDS-treated breast milk remains unchanged. 0.1% SDS can be removed from human milk without altering the creamatocrit. ELISA of serum IgG (rubella) proved it remains functional in the presence of SDS and after its removal. sIgA, IgG and IgM in breast milk are conserved after SDS-treatment when measured by RIE and ITM. CONCLUSIONS: SDS (0.025%) can inactivate HIV-1 in vitro and HSV-2 in breast milk. SDS can be efficiently removed from milk samples. SDS treatment of milk does not significantly alter protein content. Antibody function in serum and levels in breast milk are maintained after treatment and removal of SDS. 0.1% SDS does not alter fat concentration in milk and energy content is conserved. SDS or related compounds may be used to prevent breast milk transmission of HIV-1.
Stoltzfus SU et al;Pediatr 49 (4 Pt 2 Suppl): 253A (2001)
/EXPL THER/ A broad-spectrum vaginal microbicide must be effective against a variety of sexually transmitted disease pathogens and be minimally toxic to the cell types found within the vaginal epithelium, including vaginal keratinocytes. /The study/ assessed the sensitivity of primary human vaginal keratinocytes to potential topical vaginal microbicides nonoxynol-9 (N-9), C31G, and sodium dodecyl sulfate (SDS). Direct immunofluorescence and fluorescence-activated cell sorting analyses demonstrated that primary vaginal keratinocytes expressed epithelial cell-specific keratin proteins. Experiments that compared vaginal keratinocyte sensitivity to each agent during a continuous, 48-hr exposure demonstrated that primary vaginal keratinocytes were almost five times more sensitive to N-9 than to either C31G or SDS. To evaluate the effect of multiple microbicide exposures on cell viability, primary vaginal keratinocytes were exposed to N-9, C31G, or SDS three times during a 78-hr period. In these experiments, cells were considerably more sensitive to C31G than to N-9 or SDS at lower concentrations within the range tested. When agent concentrations were chosen to result in an endpoint of 25% viability after three daily exposures, each exposure decreased cell viability at the same constant rate. When time-dependent sensitivity during a continuous 48-hr exposure was examined, exposure to C31G for 18 hr resulted in losses in cell viability not caused by either N-9 or SDS until at least 24 to 48 hr. Cumulatively, these results reveal important variations in time- and concentration-dependent sensitivity to N-9, C31G, or SDS within populations of primary human vaginal keratinocytes cultured in vitro. These investigations represent initial steps toward both in vitro modeling of the vaginal microenvironment and studies of factors that impact the in vivo efficacy of vaginal topical microbicides.
PMID:10858360 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC89991 Krebs FC et al; Antimicrob Agents Chemother. 44(7): 1954-60 (2000)
3. 3= Moderately toxic: probable oral lethal dose (human) 0.5-5 g/kg, between 1 oz & 1 pint (or 1 lb) for 70 kg person (150 lb). /Alkyl sodium sulfates/
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-274
SLS is used as a surfactant in shampoos and toothpastes. SLS also has microbicidal activities against both enveloped (Herpes simplex viruses, HIV-1, Semliki Forest virus) and nonenveloped (papillomaviruses, reovirus, rotavirus and poliovirus) viruses, although it has not been approved for this use.
SLS is an anionic surfactant. Its amphiphilic properties make it an ideal detergent.
Surface-Active Agents
Agents that modify interfacial tension of water; usually substances that have one lipophilic and one hydrophilic group in the molecule; includes soaps, detergents, emulsifiers, dispersing and wetting agents, and several groups of antiseptics. (See all compounds classified as Surface-Active Agents.)
Like other surfactants, SLS is amphiphilic. It thus migrates to the surface of liquids, where its alignment and aggregation with other SLS molecules lowers the surface tension. This allows for easier spreading and mixing of the liquid. SLS has potent protein denaturing activity and inhibits the infectivity of viruses by by solubilizing the viral envelope and/or by denaturing envelope and/or capsid proteins.
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 1 companies offering 05030_FLUKA
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
